- Introduction
- Three Major Actions
- The Food Quality Protection Act (FQPA)
- Glyphosate/Roundup Case Study
Introduction
Pesticide regulators strive to prevent “unreasonable adverse effects on man and the environment,” the basic standard embedded in the FIFRA statute (the Federal Insecticide, Fungicide, and Rodenticide Act), our nation’s major pesticide regulatory law.
It is a difficult task. The science of risk assessment often lags by a decade or more the real-world risks posed by pesticides. Risks acceptable to farmers trying to protect their crop from suffocating weeds or voracious insects understandably differ from those most parents hoping to raise healthy kids are willing to accept.
The data requirements, risk-assessment protocols, and criteria supporting decisions made by the EPA’s Office of Pesticide Programs (OPP) change over time. To varying degrees, changes have tracked scientific advances in the underlying residue chemistry, toxicology, exposure, pest management, and environmental sciences.
Over time, product-specific regulations change to adjust when, where and how a pesticide can be applied. Changes also are introduced from time to time by pesticide manufacturers in the proprietary mix of co-formulants in an end-use pesticide product, altering both the environmental fate and toxicity of the pesticide. (An “end-use” pesticide product is one that is ready for sale to, and use by a pest manager).
The interaction of: (a) risk-assessment science, (b) the quantity and quality of real-world data risk assessors have access to, and (c) regulatory law and policy determines which pesticides get on the market, where and how they can be used, and who will reap the benefits and incur the costs and risks arising as a result.
Despite efforts to assure regulatory decisions are driven by legal requirements and science, politics and economics also come into play at several stages in the process, in both overt and covert ways.
Three Major EPA Actions and Decisions

The Office of Pesticide Programs (OPP) makes three types of major regulatory decisions on each new pesticide that comes on the market:
-
- Gaining Access to the Market. A “yes”/”no” response, or conditional “yes,” to an application from a pesticide manufacturer for a registration (i.e. label) authorizing a new pesticide product for use on specified crops under defined conditions, or adding a crop or other uses on the label for a pesticide already on the market.
- Losing Market Access — Imposition of Risk-Driven Cancellations or Restrictions. Cancellation, suspension, or refusal to re-register an existing registration for a pesticide already on the market for some or all uses, or re-registration of an existing product under different, and usually restricted terms and conditions.
- Establishing Tolerances — Legally Sanctioned Residues in Food. A “yes”/”no” response, or a temporary or conditional “yes,” to a petition from a pesticide manufacturer asking the EPA to establish new tolerance(s) covering residues of a pesticide in one or several foods and/or food forms (e.g., wheat grain, wheat flour, wheat germ, wheat bran).
Each of these three major actions arises from well-defined OPP/EPA application, review, risk assessment, and approval/denial processes. These processes typically require companies to conduct and submit to EPA new residue chemistry, exposure, environmental fate, and toxicology studies.
When all or most of the requested and/or required information is submitted to OPP, a scientific review is undertaken of submitted studies and OPP staff determine whether:
- All required studies have been undertaken and submitted,
- The studies were conducted in accord with Good Laboratory Practices (GLP) and other requirements set forth in EPA guidance and policy documents, and
- Study results “support” the OPP action(s) or decision(s) requested by the applicant and pesticide registrant.
Many devils lurk in the details of whether the data submitted by pesticide manufacturers “support” EPA approval of the actions requested. In the OPP, the word “support” means the submitted data is sufficient to identify possible toxicological and environmental risks, and is also sufficient to allow OPP/EPA to quantify possible risks stemming from the uses of a pesticide as proposed on its label.
The data must also be of sufficient quality and breadth to convince the EPA that the estimated levels of risks do not exceed EPA’s “level(s) of concern.”
EPA’s “level of concern” for humans, birds, or bees is driven by whether estimated risks are above or below a quantitative threshold that varies across organisms and by type of risk. In the case of humans, such risk thresholds generally assure that EPA’s best estimate of high-end exposures to people is 100-fold lower than both the pesticide’s acute and chronic Reference Doses (aRfD or cRfD).
A pesticide’s aRfD and cRfD are key human-exposure thresholds that are set by the EPA, based on its review of a pesticide’s toxicology dataset. (A cRfD becomes a chronic Population Adjusted Dose [cPAD], when an added safety factor has been applied in accord with the provisions of the 1996 Food Quality Protection Act (FQPA) (for more on the FQPA see the next section below).
Both cRfDs and cPADs establish the maximum amount of a pesticide a person of known weight can be exposed in a day, on average over a long period of time, while still assuring a “reasonable certainty of no harm,” the basic risk-standard embedded in the FQPA.
Pesticide cRfDs are set based on the active ingredient only, ignoring co-formulants or adjuvants, and are expressed in milligrams of pesticide that can be ingested per day, per kilogram of bodyweight (mg/kg/day) of the exposed person.
While a seemingly straightforward concept, the establishment of a pesticide’s cRfD or cPAD is one of the most complicated and important public-health actions undertaken across the federal government. Afterall, setting these exposure benchmarks sanctions the routine presence of poisons into the food supply.
The 1996 Food Quality Protection Act (FQPA)

The FQPA is one of the most significant public health laws passed in the last half century. It fundamentally changed the statutory standard governing the setting of pesticide tolerance levels.
It replaced the cost-benefit standard in the Federal Insecticide, Fungicide, and Rodenticide Act (FIFRA) with a solely health-based one — “reasonable certainty of no harm.”
Moreover, the FQPA provided the Environmental Protection Agency (EPA) important new authorities — and an unambiguous mandate — to assure that pregnant women, infants, and children were protected against pesticide exposures that might possibly disrupt normal pregnancies, impair the development of children, or increase the frequency and/or severity of birth defects.
Conflicting Statutes Tie EPA in Knots
From the early 1970s through July, 1996 the EPA searched for ways to navigate around conflicting statutory provisions in the two federal laws governing the establishment, review, and modification of pesticide tolerances.
Approving tolerances is the first, major gateway action the EPA must complete when the agency is asked to approve registration of a new pesticide. This is because the FIFRA requires necessary tolerances to be in place, or “on the books,” before the EPA can approve an application to register a new, food-use pesticide.
“Tolerances” are set for every food on which a pesticide can be legally applied. Pesticide labels set forth how, where (i.e. on what crops), and in what ways a pesticide can be applied. In the world of pesticides, “the label is the law.”
Tolerances work like a speed limit on the highway. Tolerance levels determine how much of a given pesticide can legally be in a particular food, while assuring a “reasonable certainty of no harm.”
Residues above the tolerance level in a given food render the food “adulterated” under the Food, Drug and Cosmetic Act (FDCA). Adulterated food is not supposed to be sold nor consumed, but often is.

The key reforms embedded in the FQPA were designed to resolve the so-called “Delaney Paradox.” The “paradox” arises from the fact that a provision in one federal law (the Delaney Clause in the FDCA) intended to reduce cancer risk was actually, in some cases, increasing it, because of the provisions in another federal law (FIFRA).
How could this happen?
Pesticide manufacturers and farmers typically push EPA to place heavy weight on the economic benefits of all pesticides, including cancer-causing ones, thereby blocking EPA actions intended to restrict or end the use of high-risk pesticides. They are often successful in doing so, especially when there were few, or no similarly effective pesticide alternatives on the market.
Along comes a pesticide company with an effective, alternative, newly discovered pesticide, one that would give farmers a badly needed option, and reduce the benefits associated with the ongoing use of a cancer-causing pesticide already on the market.
EPA approval of the new pesticide would lead to a downward adjustment in the benefits stemming from future use of the older, cancer-causing pesticide, and would presumably accelerate EPA actions to drive it off the market or curtail its use.
But sometimes the new pesticide might pose low, or very low cancer risks. Then, because of the Delaney Clause, the new pesticide would not be approved by the EPA, even in cases where the new pesticide posed 1/100th or less of the cancer risk stemming from applications of the older pesticide it would replace.
Unraveling the Delaney Paradox
The statutory and regulatory roots of the Delaney Paradox are complex. In short, the anti-cancer Delaney Clause applies with no wiggle room for EPA in registering new pesticides, but plays no role when the EPA is deciding whether to cancel or restrict the future use of an already-registered, oncogenic pesticide.
In short in OPP under then-existing federal law, new pesticides were guilty until proven innocent of posing excessive risks, while older pesticides on the market were innocent until EPA could prove they pose excessive risks — and with EPA bearing a heavy burden of proof that often was beyond the reach of risk-assessment science.
The legislative debate over the reform of the Delaney Clause was protracted, controversial, and closely followed. Each year from the the 1980s through 1996, conflicting legislative proposals were introduced by members of Congress supportive of either stricter application of the Delaney Clause, or the repeal of the Clause itself.
The high drama over the reform of the Delaney Clause came to a surprisingly abrupt end on July 24, 1996, the day the House of Representatives passed the Food Quality Protection Act (FQPA) without a single dissenting vote after only a few minutes of floor discussion.
On the next day, the Senate passed the bill on a unanimous consent motion after barely a minute of discussion.
Four days later in its Sunday edition, the Washington Post ran a lengthy story describing the long process that led to the FQPA and explaining the bill’s major provisions. The lead paragraph in that story states –
“The new federal food safety law, which swept through Congress without opposition and was blessed by many industry and environmental groups, is a rare legislative compromise in which all sides can declare a measure of victory.” (Washington Post, July 28, 1996).

In his August 3rd statement at the FQPA signing ceremony, President Bill Clinton said:
“From the day I took office I have worked hard to meet what I think is a fundamental promise that we should make to our people. People should know that the food they eat and the water they drink will not make them sick…
“Today we add the cornerstone to this solid foundation [of new laws] with the Food Quality Protection Act. I like to think of it as the ‘peace of mind’ act, because it’ll give parents the peace of mind that comes from knowing that the fruits, the vegetables, the grains that they put down in front of their children are safe. It’s long overdue. The old safeguards that protected our food from pesticides were written with the best of intentions, but they weren’t up to the job. And as you can see from the vast array of support here across every sector of American life, nobody liked them very much and no one thought that they really worked as they were supposed to. Bad pesticides stayed on the market too long, good alternatives were kept out.”
Core Provisions

The FQPA incorporated into federal law the major recommendations of the 1987 NAS Delaney Paradox report, as well as the 1993 NAS report Pesticides in the Diets of Infants and Children (NAS, 1993).
A new and consistent standard – “reasonable certainty of no harm” – was put in place to govern the review, establishment, and adjustment of all pesticide tolerances.
The EPA was directed to place greater weight on the risks faced by pregnant women, infants, and children.
New provisions were added to FIFRA to transform the statute’s risk-benefit balancing decision rule to a purely health-based standard for the purpose of tolerance setting.
All petitions for new pesticide tolerances were to be reviewed and approved in accord with the new “reasonable certainty of no harm” standard, and the approximate 9,700 tolerances in place at the time were to be systematically reviewed, and when needed, adjusted or revoked.
Plus —
The EPA was directed to impose an additional 10-fold safety factor in setting each pesticide’s chronic Reference Dose, unless the Administrator of EPA determines that the agency has thorough and adequate studies to evaluate developmental and reproductive risks, and deems such risks as consistent with the FQPA’s “reasonable certainty of no harm” standard.
All future regulatory decisions must be based on aggregate exposures to a given pesticide via the diet, drinking water, dermal, inhalation, and any other routes of exposure. Prior to the FQPA, EPA made safety judgements on tolerances based just on residues in food, and decisions on occupational exposures based just on work-related exposures, despite the fact most people are exposed to pesticide through several different routes at various stages of life.
In one of the most innovative and important reforms, the FQPA directed the EPA to conduct cumulative risk assessments taking into account the sum or aggregate exposures to all pesticides that pose risks to people through the same mode of biological action.
This is the provision of the FQPA that forced EPA to conduct its first-ever, cumulative risk assessment of the organophosphate class of insecticides, all of which are cholinesterase inhibitors and developmental neurotoxins.
Impacts of the FQPA
Later in 2019, Hygeia Analytics will be releasing a detailed quantitative accounting of the impact of the FQPA over the last 22 years. In short, there is some good news, but also many missed opportunities.
Pronounced shifts have occurred since 1996 in the distribution of pesticide dietary risks across families of pesticide chemistry, between domestically grown and imported food, and between conventionally grown and organic food.
Overall risks have declined by about two-thirds in most domestically grown foods that make up important parts of the diets of infants and children.
Serving-for-serving, pesticide dietary risks stemming from residues in domestically grown fruits and vegetables have fallen, in contrast to residues and risk stemming from imported foods, which have often risen.
A relatively small number of food-pesticide combinations accounts for a highly disproportionate share of total dietary risks. In most crops, one to three risk-driver pesticides account for 90 percent or more of aggregate dietary risk. A single risk driver accounts for 80 percent or more of aggregate dietary risk in around one-half of major fresh fruits and vegetables.
So by targeting restrictions to just relatively high-risk pesticide-food combinations, the EPA could dramatically reduce pesticide dietary risk, without major impacts on the pesticides available to farmers.
The EPA has imposed significant regulatory restrictions on around a dozen, major risk-driver pesticides (most of them organophosphate insecticides). Other risk drivers persist in children’s foods that have yet to be impacted significantly by EPA actions.
EPA regulatory restrictions and/or tolerance reductions have impacted around one-third of the most serious risk drivers. Dozens of other pesticide-food combinations have emerged, and more will emerge in the future as risk drivers, because of the lack of change in old, excessively high tolerances that remain on the books, despite being out of step with the FQPA’s “reasonable certainty of no harm” standard.
Glyphosate/Roundup Case Study
Introduction
Since the introduction of genetically engineered, glyphosate-resistant crops in 1996, this broad-spectrum herbicide quickly rose to become the most heavily used pesticide in the world, ever. By 2014, U.S. farmers were applying around 250 million pounds of glyphosate a year (see chart below).

The Lowdown on Roundup
For the more information on glyphosate development, regulation, and use, see our Lowdown on Roundup presentation series — Part I: Discovery and Commercialization, Part II: Regulatory Milestones, and Part III: Glyphosate Use.

Changes Over Time
The “Lowdown on Roundup” describes how overall glyphosate use has risen dramatically since 1996, the year the first crops went on the market that were genetically engineered to allow “over-the-top,” post-emergence applications of Roundup for full-season weed control.
The 2016 paper “Trends in Glyphosate Use in the United States and Globally” covers in detail trends in glyphosate use both in the U.S. and globally. It covers both agricultural and non-ag use of glyphosate-based herbicides, and isolates the impact of the three major GE crops — soybeans, cotton, corn — on glyphosate and overall herbicide use. Glyphosate use has risen almost 15-fold since the introduction of Roundup Ready crops in 1996. Genetically engineered crops account for about 56% of global glyphosate use.
Recently a retired Bayer scientist based in the U.K. published a paper in Outlooks on Pest Management on the impact of GE crops on glyphosate use. It states in part:
“Glyphosate use in these HT crops has clearly increased compared to the non-selective uses alone prior to the commercialisation of crops tolerant to this herbicide. The magnitude of this is illustrated by Benbrook (2016) who has compiled the use data for glyphosate both in the USA and globally. In 2014, virtually all cotton and soybean acreage in the USA was treated with glyphosate as was 77% of maize acreage.”
–Ken Pallet, Bayer CropScience (retired) in “Engineered Crop Tolerance to Glyphosate and its Impact on the Use of the Herbicide,” from the December 2018 issue of Outlooks on Pest Management
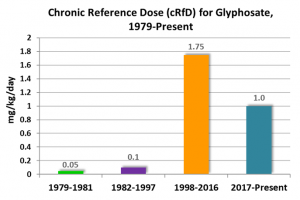
In estimating the human health risks arising from applications of glyphosate-based herbicides, regulators have to set a key exposure threshold called the chronic Reference Dose (cRfD). This exposure threshold determines how much glyphosate a person of known weight can be exposed to in a day, without exceeding EPA’s chronic exposure “level of concern.”
The figure below shows how glyphosate’s chronic reference dose (cRfD) has changed over time, in response to the receipt of new studies and EPA re-assessments of the glyphosate toxicological database.
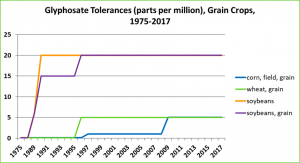
There have also been many changes in glyphosate tolerances in key food crops including corn, soybeans, and wheat, as shown in the chart below. The increases in wheat, and soybean tolerances have been driven by EPA approval of pre-harvest, desiccation uses of glyphosate-based herbicides. Such pre-harvest uses lead to greatly elevated residues in harvested wheat and soybean grain because of the short time between a direct application onto an almost-ready to harvest grain crop.
Does Glyphosate/Roundup Cause Cancer?

Seemingly similar pesticide risk assessments can come to different conclusions depending on what information is considered, and how much weight is placed on different lines of evidence by regulators.
Two factors usually play a role in explaining differences in regulatory assessments when they arise: (1) whether, and to what extent risk assessments took into account exposures to formulated pesticide products, versus data on just active ingredients, and (2) the degree to which studies published in peer-reviewed journals are considered in the evaluation process, in addition to studies conducted by registrants and submitted to regulatory agencies.
A key example of the interplay of changing science and regulatory policy in pesticide regulation is the question of whether glyphosate, the most heavily used pesticide in the world, should be classified as a carcinogen.
The widely respected International Agency for Research on Cancer (IARC) has concluded that GBHs are “probably carcinogenic to people,” while the U.S. EPA asserts that glyphosate likely does not pose a cancer risk to people based on typical levels of dietary exposure. Why did EPA and IARC reach such different conclusions?
Differences in the IARC and EPA assessments of the genotoxicity of glyphosate and GBHs lie at the heart of the differing cancer judgement reached by IARC and EPA, as explained in the paper “How did the U.S. EPA and IARC reach diametrically opposed conclusions on the genotoxicity of glyphosate-based herbicides?” in the peer-reviewed journal Environmental Sciences Europe. The paper and related information is accessible here.
Other Resource
- More on human health impacts of pesticides
- Buddhini Samarasinghe’s excellent informational article “Hallmarks of Cancer” was published in “Scientific American” in 2013-2014 and explains how cancer develops.
- 2019 papers in the American Journal of Public Health on the risk assessment of glyphosate.
- Morabia, A.; “Fighting Independent Risk Assessment of Talc and Glyphosate: Whose Benefit Is It Anyway?;” American Journal of Public Health, 2019, 109(7), 955-956; DOI: 10.2105/AJPH.2019.305144.
- Vineis, P.; “Public Health and Independent Risk Assessment;” American Journal of Public Health, 2019, 109(7), 978-980; DOI: 10.2105/AJPH.2019.305142.
- Samet, J. M.; “Expert Review Under Attack: Glyphosate, Talc, and Cancer;” American Journal of Public Health, 2019, 109(7), 976-978; DOI: 10.2105/AJPH.2019.305131.
